Sample Management Program
is a comprehensive digital solution designed to manage the entire lifecycle of samples efficiently a...
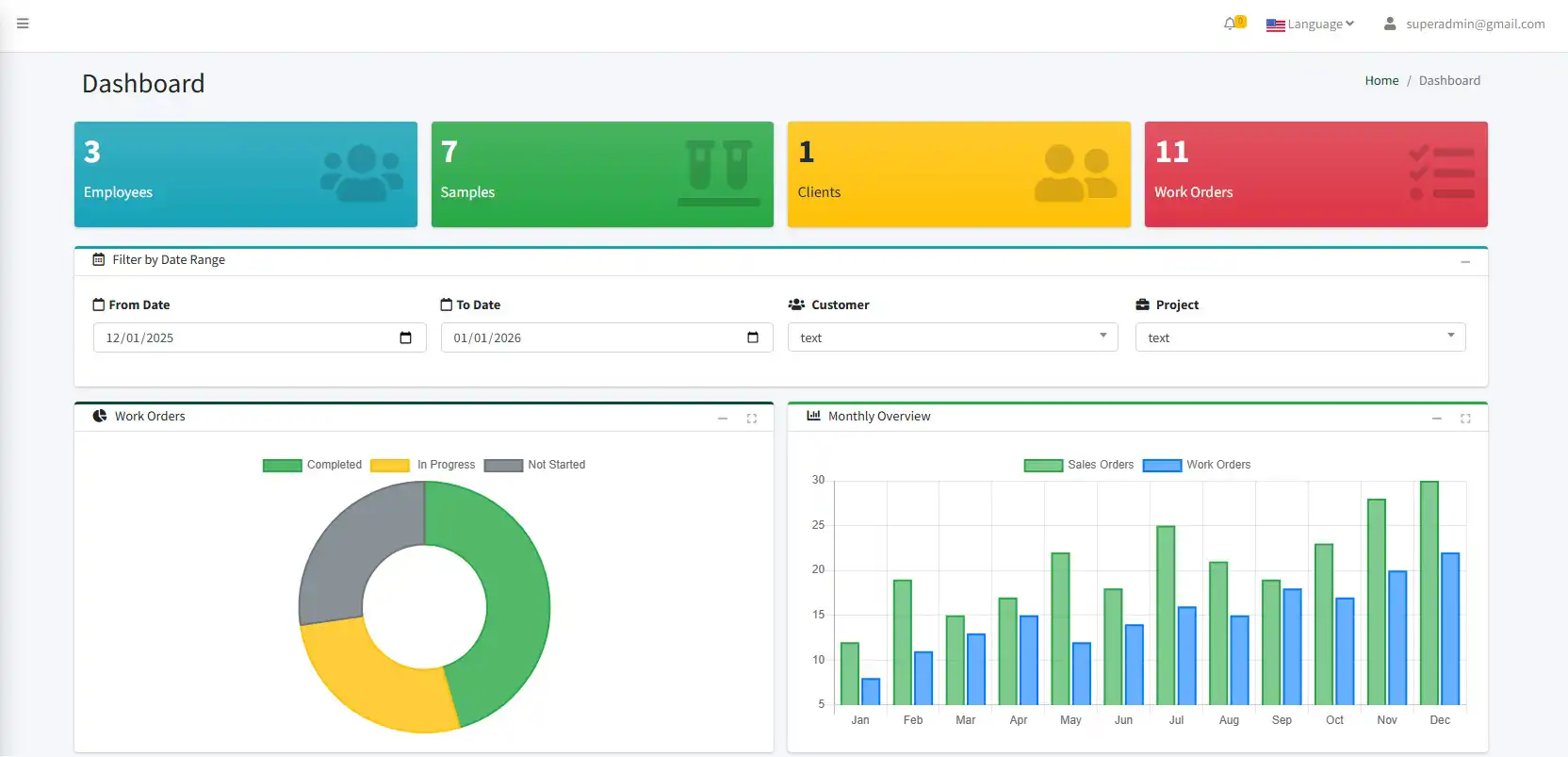
is a comprehensive digital solution designed to manage the entire lifecycle of samples efficiently and accurately. From sample registration and reception to processing, analysis, and final reporting, the system ensures that every step is properly recorded and controlled. It provides full traceability of samples in real time, enabling users to monitor status, location, and handling history at any stage. The program reduces manual errors by standardizing workflows and automating routine tasks. With secure data management and role-based access, it guarantees data integrity and confidentiality. Ultimately, the system improves operational efficiency, enhances data reliability, and supports informed decision-making.
The program enables real-time sample tracking, allowing users to monitor the status, location, and responsibility of each sample at any stage. By maintaining a complete audit trail, it enhances transparency, traceability, and compliance with quality and regulatory standards. All sample-related activities are automatically logged, providing a clear history of actions and changes. This ensures accountability across teams and simplifies internal audits and inspections. The system also supports standardized procedures, helping organizations meet industry regulations with confidence. As a result, operational risks are reduced and overall quality management is strengthened.